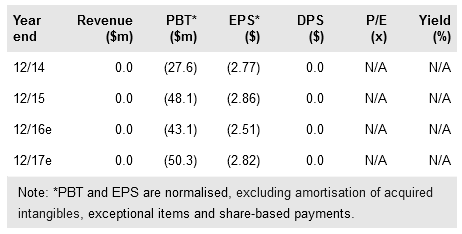
Tonix Pharm (NASDAQ:TNXP) reported positive Phase II results for TNX-102 SL (sublingual formulation of cyclobenzaprine) marking the first-ever large multicenter trial for military-related PTSD. The 237-person study compared two dosage levels (2.8mg and 5.6mg) against placebo and found a significant improvement for the 5.6mg dose (p=0.038) in the Clinician-Administered PTSD Scale for DSM-5 (CAPS-5), the primary endpoint for approval.
Significant improvement across multiple metrics
As part of the CAPS-5 assessment, the 5.6mg dose of TNX-102 SL showed significant improvement in measures of hyperarousal (p=0.048), sleep disturbances (p=0.01) and startle response (p=0.015). These clinical manifestations translated into improved physician (p=0.041) and patient (p=0.035) global assessments, as well as improved function at work (p=0.05) and in social situations (p=0.031).
To read the entire report Please click on the pdf File Below