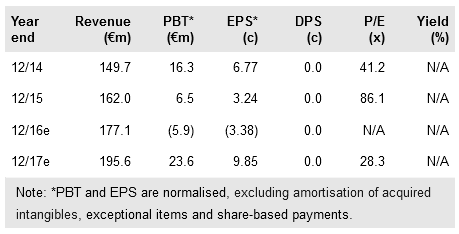
Positive results from Pharma Mar's (MC:PHMR) ADMYRE Phase III trial of Aplidin in multiple myeloma clear the path for a potential approval in Europe in H217. Separately, PharmaMar continues to accelerate clinical development of PM1183, a follow-on to its marketed anticancer drug Yondelis: a Phase III trial of PM1183 in small cell lung cancer planned for mid-year will add to the ongoing CORAIL Phase III in ovarian cancer (an interim futility analysis after 210 ovarian patients is expected to report in H216). Our valuation increases to €1.09bn (was €1.07bn previously) or €4.91 per share (€4.82), but a successful self-commercialisation of PM1183 in the US (if approved) could lift valuation by 14% compared to our base case.
Aplidin: Positive results in ADMYRE pivotal trial
The 255-patient ADMYRE Phase III trial of Aplidin in relapsed/refractory multiple myeloma met its primary endpoint last month, showing a statistically significant 35% reduction in the risk of disease progression or death. PharmaMar plans to file for approval in Europe in Q416, which could potentially lead to an approval in H217 and market launch in early 2018. With US approval likely to depend on the forthcoming pivotal trial in T-cell lymphoma, we delay our forecast US launch date for Aplidin by three years to 2021.
To read the entire report Please click on the pdf File Below