Maiden pre-tax profit and a broadening pipeline
Improving margins for Imvamune supply to the US government helped deliver Bavarian Nordic's (BAVA.COP) first pre-tax profit in FY13. Further deliveries are on track for 2014 and two of three Imvamune trials have initiated. However, completion of enrolment in the Phase III PROSPECT trial of main value driver Prostvac for prostate cancer is now expected by year end. Country-specific regulatory delays have affected clinical site opening in Germany and the Netherlands, but this is offset by initiatives in other regions to boost recruitment. We believe the first futility analysis should happen in 2014. Pipeline progress will see other assets start clinical trials this year.
Cancer pipeline: Progress expected in 2014
PROSPECT delays have no impact on expected final data read (H216); first interim analysis may occur this year; efficacy analyses are likely in 2015. FDA feedback on CV-301 development in colorectal cancer is expected in H214; Ph II is contingent on funding. Two NCI-sponsored trials will initiate in H114: Phase II CV-301 bladder cancer and Phase I advanced cancer with new project, MVA-BN Brachyury.
Infectious diseases: Steady margin improvement
Investment in production has yielded improved Imvamune margins: now 60.9% on a rolling 12-month basis (64.7% in Q413). The final $118m of the April 2013 US government contract should be secured in 2014. The US market is the main Imvamune value driver (the first of two Phase II trials for BLA filing has started), although orders from European and Canadian governments are anticipated.
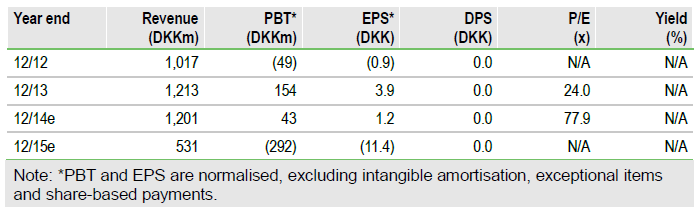
Financials: Targeting EBIT breakeven for FY14
FY13 results exceeded management guidance: revenue was DKK1.2bn (DKK1.1bn guided) with PBT of DKK6m (break even). Cash preparedness (gross cash plus undrawn DKK120m credit line) was DKK652m (vs DKK600m). New FY14 guidance is for revenue of DKK1.2bn, EBIT break even and cash preparedness of DKK600m.
Valuation: DKK2,944m with upside potential
Our DCF-based valuation of DKK2,944m, DKK113/share (formerly DKK2,965m, DKK114/share) reflects increased 2014 R&D spend and a US$/DKK5.4 FX rate offset by a higher cash balance. Positive data from the Prostvac PROSPECT study (2016) and/or interim analysis (H214 onwards); partnering deals for Prostvac and CV-301 in CRC; first confirmed European orders for Imvanex (2014/15); and FDA filing of Imvamune (2016) would represent valuation upside.